Formula Units
Pure substances and chemical formula
- Atoms are the small building blocks of matter
- One of the broadest classifications of matter are pure substances
- Pure substances can be classified into elements or compounds
- Elements are composed of only one kind of atom
- Aluminum (Al) or oxygen (O2)
- Compounds have a unique combination of atoms and they can be split into molecular or ionic
- This combination of atoms can be represented with a chemical formula, in which atomic symbols identify the elements in the compound and subscripts are used to show how many atoms are present
Molecular compounds
- A molecular compound is composed of individual molecules.
- A molecule is formed when two or more atoms are bonded together with a covalent bond
-
- Water is a molecular compound because its atoms are bonded with covalent bonds
- The chemical formula of water is H2O
A molecule of water (H2O)

A molecule of water (H2O) showing 2 hydrogen atoms, 1 oxygen atom and the covalent bonds
Ionic compounds
- An ionic compound consists of atoms or ions held together in a formula unit
- A formula unit is the arrangement of atoms or ions in fixed proportions by an ionic bond
- Salt (sodium chloride) is an ionic compound because its ions are held together in a fixed arrangement by ionic bonds
- The chemical formula of sodium chloride is NaCl
Formula unit of sodium chloride (NaCl)
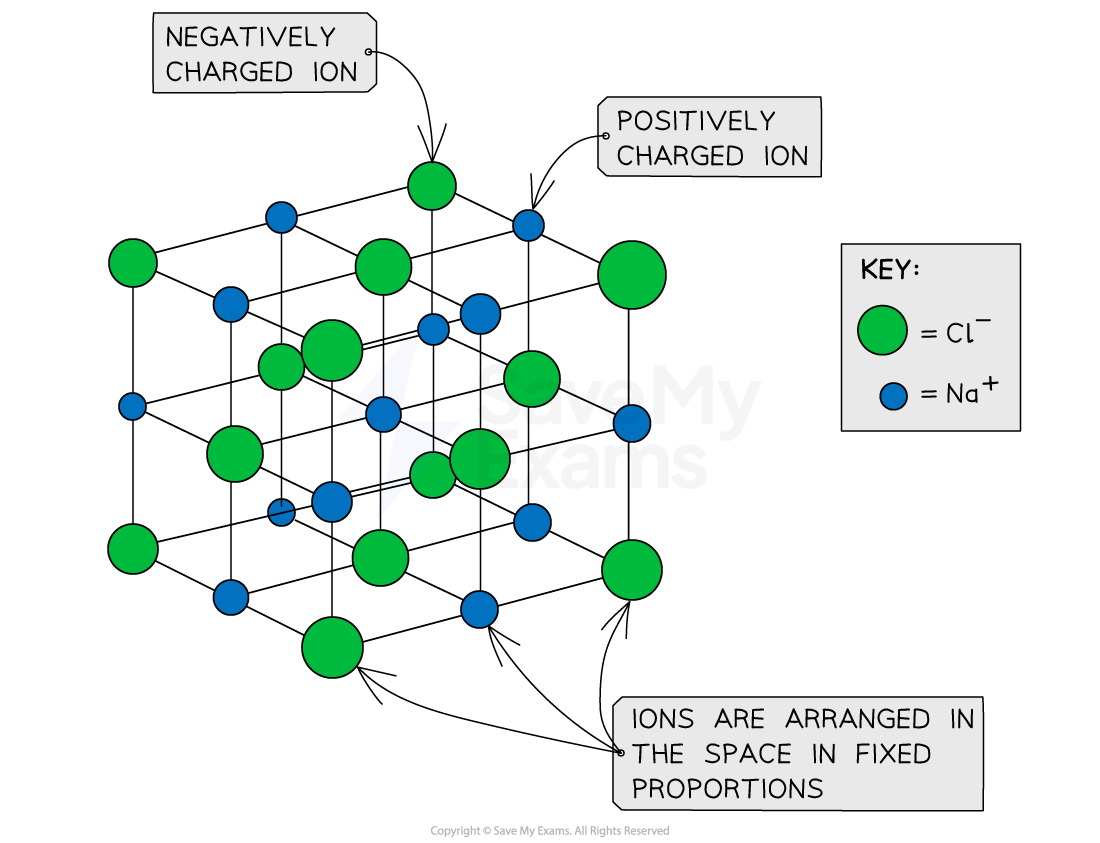
A formula unit of sodium chloride (NaCl) showing the regular but alternating arrangement of sodium ions and chloride ions


